Review – OPEN ACCESS
Ahmed Edris, Silke De Feyter, Tania Maes, Guy Joos & Lies Lahousse
Respiratory Research volume 20, Article number: 179 (2019)
Abstract
Since novel treatments to target eosinophilic inflammation in Type 2 asthma are emerging, we aimed to evaluate and meta-analyze the efficacy of monoclonal antibodies to reduce exacerbation rate. PubMed and Web of Science were searched for phase II and phase III randomized clinical trials with monoclonal antibodies targeting key mediators of type 2-associated asthma. Thirty trials were selected involving biologics that target the IL-5 pathway, IL-13, the common IL-4 and IL-13 receptor, IL-9, IL-2 and TSLP. As no head-to-head trials were retrieved from literature, we performed an arm-based network meta-analysis to compare effects on exacerbation rate between the different treatments.
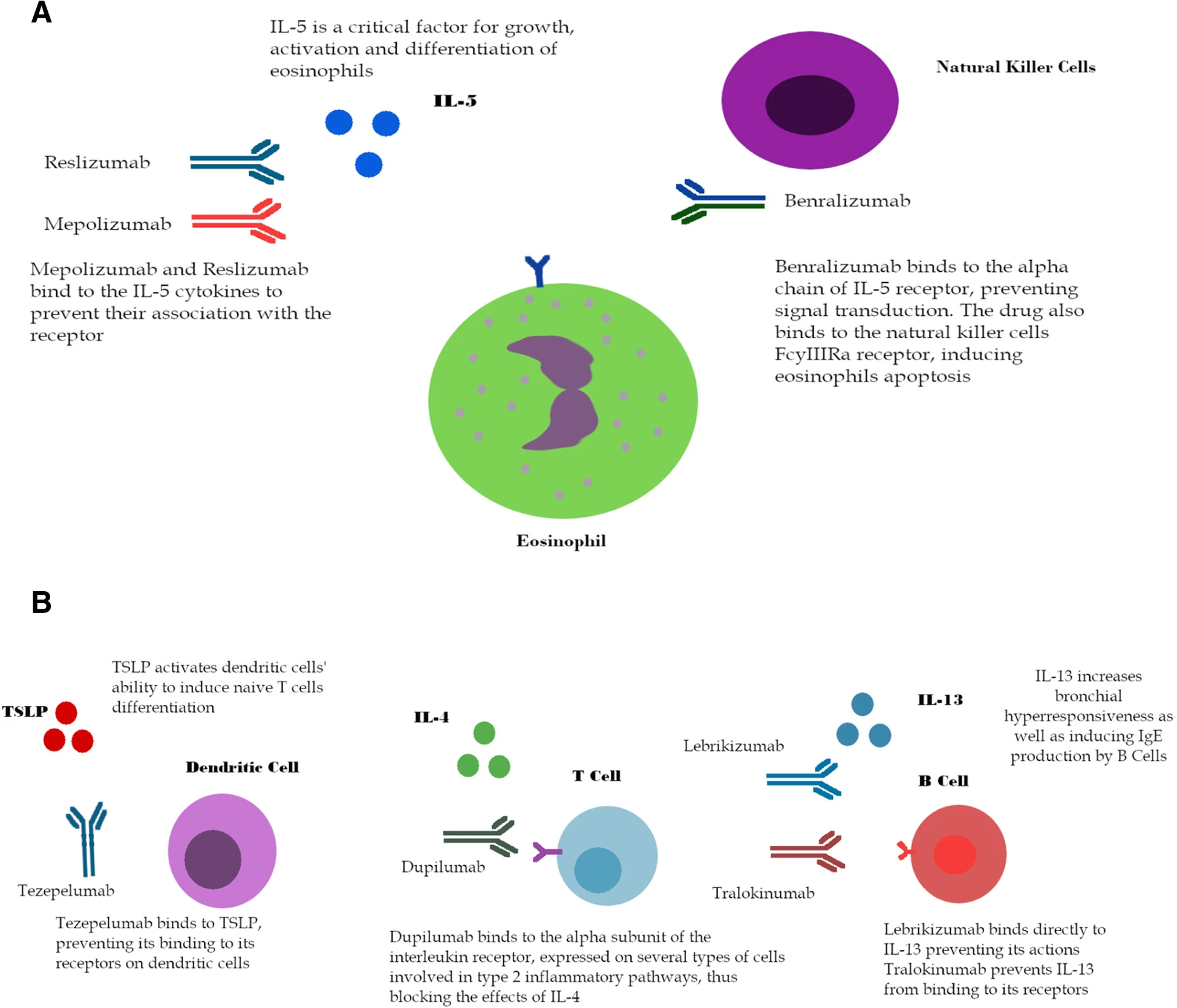
Mepolizumab, reslizumab and benralizumab significantly reduced the risk of exacerbations compared to placebo (by 47–52%, 50–60%, and 28–51% respectively). Reslizumab and benralizumab also improved lung function. Dupilumab and tezepelumab improved lung function in frequent exacerbators. Lebrikizumab had no significant effect on the number of exacerbations, symptom control or health-related quality of life. Tralokinumab improved lung function compared to placebo. Network meta-analysis of all treatment and placebo arms, showed no superiority of one biologic over the others. Large reductions in exacerbation rates were observed compared to placebo, though only benralizumab was sufficiently powered (n = 2051) to demonstrate significantly decreased exacerbation rates in the subgroup analysis of IL-5 acting agents compared to placebo.
Monoclonal antibodies such as mepolizumab, reslizumab and benralizumab have proven their benefit to reduce exacerbation rates in severe persistent eosinophilic asthma in the published trials. However, no statistically significant superiority was observed of one biologic over the other in the network meta-analysis. More studies with direct head to head comparisons and better defined endotypes are required.
