Sharma, R., Tiwari, A., Kho, A.T. et al. BMC Pulm Med 24, 553 (2024). https://doi.org/10.1186/s12890-024-03372-4
Abstract
Background
Bronchodilator response (BDR) is a measure of improvement in airway smooth muscle tone, inhibition of liquid accumulation and mucus section into the lumen in response to short-acting beta-2 agonists that varies among asthmatic patients. MicroRNAs (miRNAs) are well-known post-translational regulators. Identifying miRNAs associated with BDR could lead to a better understanding of the underlying complex pathophysiology.
Objective
The purpose of this study is to identify circulating miRNAs associated with bronchodilator response in asthma and decipher possible mechanism of bronchodilator response variation.
Methods
We used available small RNA sequencing on blood serum from 1,134 asthmatic children aged 6 to 14 years who participated in the Genetics of Asthma in Costa Rica Study (GACRS). We filtered the participants into the highest and lowest bronchodilator response (BDR) quartiles and used DeSeq2 to identify miRNAs with differential expression (DE) in high (N = 277) vs. low (N = 278) BDR group. Replication was carried out in the Leukotriene modifier Or Corticosteroids or Corticosteroid-Salmeterol trial (LOCCS), an adult asthma cohort. The putative target genes of DE miRNAs were identified, and pathway enrichment analysis was performed.
Results
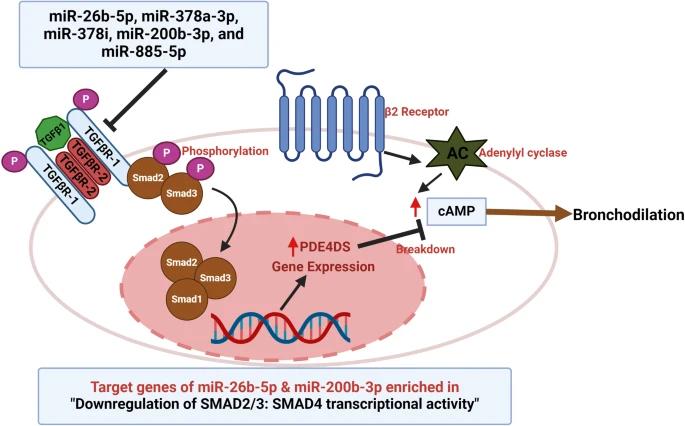
We identified 10 down-regulated miRNAs having odds ratios (OR) between 0.37 and 0.76 for a doubling of miRNA counts and one up-regulated miRNA (OR = 2.26) between high and low BDR group. These were assessed for replication in the LOCCS cohort, where two miRNAs (miR-200b-3p and miR-1246) were associated. Further, functional annotation of 11 DE miRNAs were performed as well as of two replicated miRs. Target genes of these miRs were enriched in regulation of cholesterol biosynthesis by SREBPs, ESR-mediated signaling, G1/S transition, RHO GTPase cycle, and signaling by TGFB family pathways.
Conclusion
MiRNAs miR-1246 and miR-200b-3p are associated with both childhood and adult asthma BDR. Our findings add to the growing body of evidence that miRNAs play a significant role in the difference of asthma treatment response among patients as it points to genomic regulatory machinery underlying difference in bronchodilator response among patients.
