Colby Stong
About 19% of patients with severe asthma achieved clinical remission and 79% had a clinical response after 1 year of biologic therapy, according to a study in Chest.
Investigators identified the level of clinical remission in patients with severe asthma who used biologics, as well as predictors of treatment response, using data from the Danish Severe Asthma Register (DSAR), which includes all patients treated with a biologic for severe asthma in Denmark.
Participants initiated biologic treatment between January 1, 2016, and December 31, 2021. The analysis included 501 patients (mean [SD] age, 56 [14] years; 51% female). Patients were at least 18 years of age, except for 1 aged 16 and 1 aged 17 years.
A clinical response was defined as having a decrease of at least 50% in annualized exacerbation rate if the indication was based on at least 2 exacerbations in the preceding 12 months before treatment, and a reduction of at least 50% in oral corticosteroids (OCS) dose from baseline if the indication was based on the need for maintenance OCS (mOCS). Clinical remission was regarded as a complete absence of exacerbations and need for mOCS and well-controlled symptoms (6-question Asthma Control Questionnaire [ACQ-6] score ≤1.5) after 12 months of treatment.
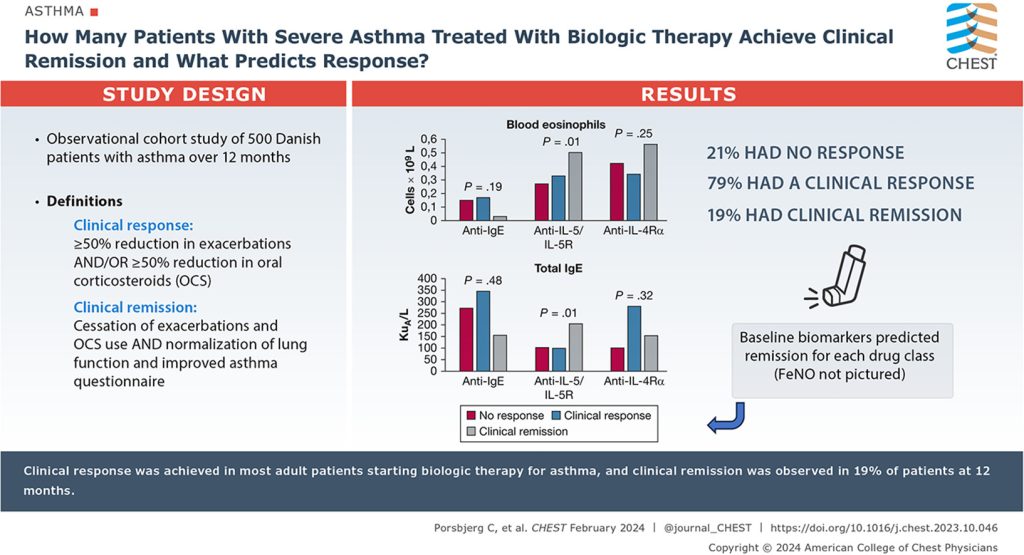
After 12 months, 397 (79%) participants had a clinical response, and 104 (21%) had no response to treatment. Of the clinical responders, 97 patients (24% of those with a clinical response, 19% overall) had clinical remission.
The proportion of clinical response varied among the 3 individual drug classes, from 72% who used anti-immunoglobulin E, 78% for anti-interleukin (IL)-5/IL-5R, and 92% for anti-IL-4Rα. The proportions of patients with remission in these 3 groups were 6%, 19%, and 30%, respectively.
The participants who had remission were more likely to be male, have a lower body mass index (BMI), be older at asthma onset, to not use mOCS, have a lower ACQ-6 score, have higher forced expiratory volume in 1 second (FEV1) and FEV1 percent predicted, and have a greater median blood eosinophil count. Also, they were less likely to have chronic obstructive pulmonary disease, dysfunctional breathing, and nasal polyposis.
The strongest predictors of remission in the multivariate model were BMI (odds ratio [OR] for 1-unit increase, 0.91; 95% CI, 0.86-0.97) and disease duration (OR for 1-year increase, 0.98; 95% CI, 0.97-0.99). After doubling concentrations of blood eosinophil count and fractional exhaled nitric oxide, the ORs were 1.18 (95% CI, 0.98-1.42) and 1.02 (95% CI, 0.81-1.27), respectively. Male sex had an OR of 1.57 (95% CI, 0.88-2.83) vs female sex.
After 12 months of treatment, 65% of participants who had a clinical response did not have any exacerbations, and 75% of patients were not using mOCS.
Limitations include the lack of a control arm and missing patient data.
“Importantly, the chance of remission was better in patients with shorter disease duration and less severe disease, suggesting that early intervention is important for achieving optimal results,” the investigators stated.
Disclosure: The DSAR is funded by grants from GlaxoSmithKline, Novartis, AstraZeneca, Teva, and Sanofi. Some of the study authors declared affiliations with biotech, pharmaceutical, and/or device companies. Please see the original reference for a full list of authors’ disclosures.
References: Hansen S, Søndergaard MB, von Bülow A, et al. Clinical response and remission in patients with severe asthma treated with biologic therapies. Chest. 2024;165(2):253-266. doi: 10.1016/j.chest.2023.10.046
