ABSTRACT
Background
Severe uncontrolled asthma (SUA) is frequently treated with biologic therapy if a T2 phenotype is found. Bronchoscopy is not routinely recommended in these patients unless a specific indication to rule out comorbidities is present.
Research question
Is routine bronchoscopy safe and useful in phenotyping and endotyping SUA patients prior to the indication of a biologic therapy?.
Study Design and Methods
Prospective study of consecutive SUA patients who were referred to a specialized asthma clinic to assess the indication of a biologic therapy. Patients were clinically phenotyped as T2-allergic, T2-eosinophilic and non-T2. All patients underwent bronchoscopy and systematic data collection of endoscopic findings, microbiology of bronchial aspirate (BAS) and presence of eosinophils in bronchial biopsy were recorded and compared between asthma phenotypes. Cluster analysis was performed accordingly.
Results
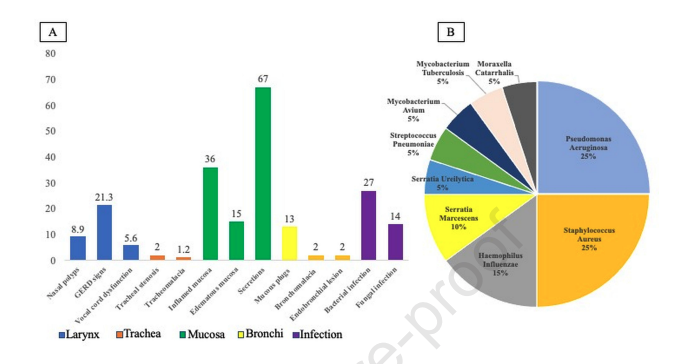
One-hundred patients were recruited and classified as T2-allergic (28%), T2-eosinophilic (64%) and non-T2 (8%). On bronchoscopy, signs of gastroesophageal reflux disease were detected in 21%, vocal cord dysfunction in 5% and tracheal abnormalities in 3%. BAS culture isolated bacteria in 27% of patients, and fungi in 14%. Three clusters were identified: non-specific, upper airway, and infection being the latter less frequently associated to submucosal eosinophilia. Eosinophils were detected in 91% of bronchial biopsies. Despite a correlation to blood eosinophils, five patients with T2-phenotypes showed no eosinophils in bronchial biopsy, and 3 patients with non-T2 showed eosinophils in bronchial biopsy. Only one patient had a moderate bleeding.
Interpretation
Routine bronchoscopy in SUA eligible for biologic therapy is a safe procedure that can help to better phenotype and personalize asthma management.
Cosío BG, Shafiek H, Mosteiro M, Iglesias A, Gómez C, Toledo-Pons N, Martinez R, Lopez M, Gimeno IE, de Llano LP. Redefining the role of bronchoscopy in the work-up of severe uncontrolled asthma in the era of biologics: a prospective study. Chest. 2023 Mar 13:S0012-3692(23)00354-9. doi: 10.1016/j.chest.2023.03.012.
